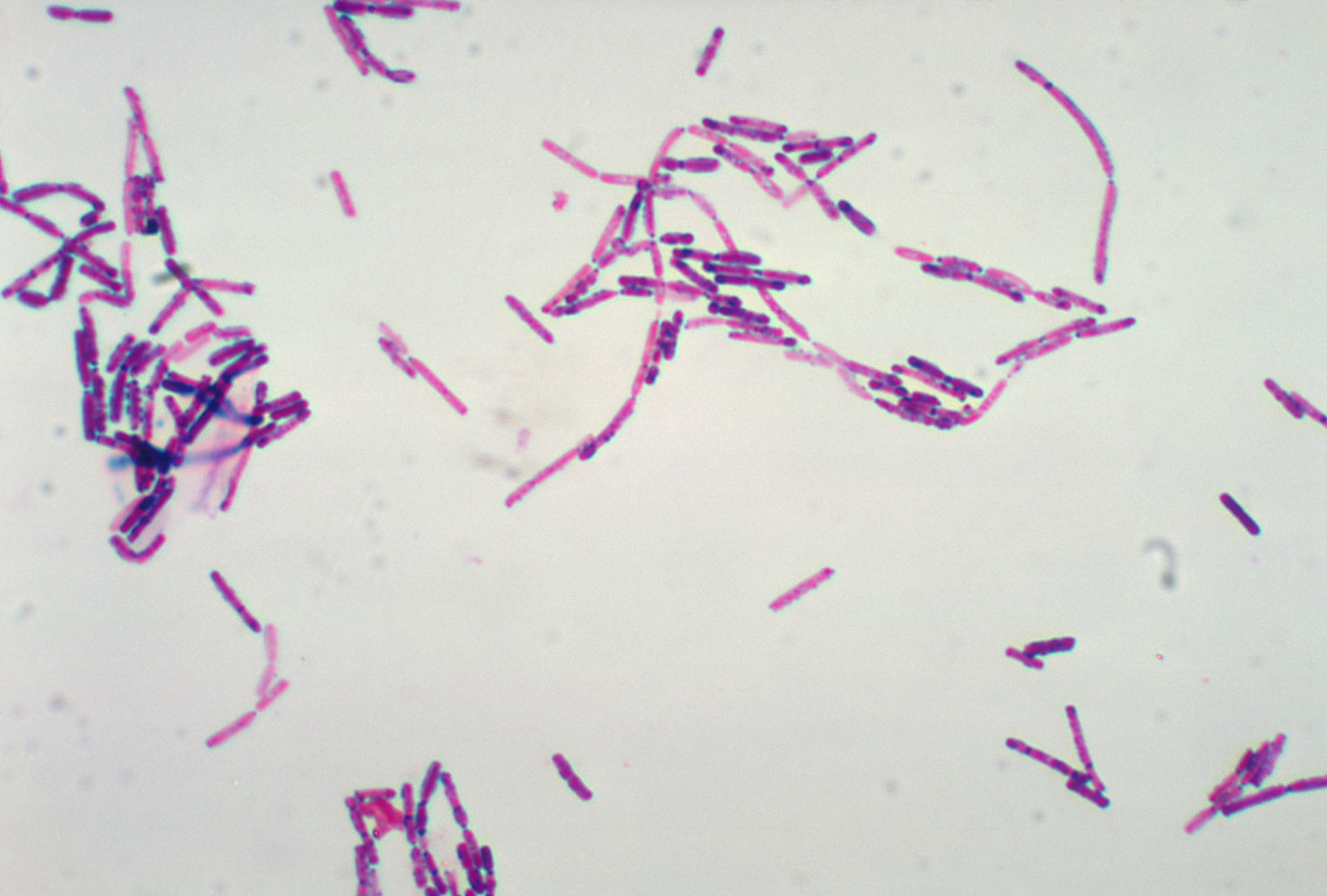
That allows the endospore to show up as red, while the rest of the cell stains blue. To combat this, a special stain technique called a Moeller stain is used. While the rest of a bacterial cell may stain, the endospore is left colorless. Viewing endospores under the light microscope can be difficult due to the impermeability of the endospore wall to dyes and stains. Figure: Endospore morphology: Variations in endospore morphology: (1, 4) central endospore (2, 3, 5) terminal endospore (6) lateral endospore. Bacteria produce a single endospore internally. They are commonly found in soil and water, where they may survive for long periods of time. They are resistant to ultraviolet radiation, desiccation, high temperature, extreme freezing and chemical disinfectants. Endospores can survive without nutrients. The endospore consists of the bacterium’s DNA and part of its cytoplasm, surrounded by a very tough outer coating. Examples of bacteria that can form endospores include Bacillus and Clostridium. When the environment becomes more favorable, the endospore can reactivate itself to the vegetative state. Endospores enable bacteria to lie dormant for extended periods, even centuries. In endospore formation, the bacterium divides within its cell wall. Endospore formation is usually triggered by lack of nutrients, and usually occurs in Gram-positive bacteria. Describe the function and advantage of endospore formation, as well as the methods for viewing it.Īn endospore is a dormant, tough, and non-reproductive structure produced by certain bacteria from the Firmicute phylum.Spores appear green in red-stained cells.\).Wash with water, air dry and observe under oil immersion objective.Wash the water and counterstain with either safranine or mercurochrome for 30 seconds.Alternatively, place the slide on the boiling water beaker and stain for 10 min with malachite green. Boil malachite green in a test tube and pour it over the smears for 5-10 min.Spores are stained red and cells appear colorless against black background.Air-dry and observe under oil immersion objective. Add a drop of nigrosine and spread it on the slide with another slide. Boil ZNCF stain in a test tube and pour it over the smear and let stain for 5 min.Make smears from each culture on clean glass slide air dry and heat fix.Note the color of the spore and cell in each. Repeat the spore staining using ZNCF instead of malachite green as counterstain in lieu of safranine.Blot-dry the smear and examine under oil immersion objective. Counterstain the slide for 30 seconds with safranine.Wash the stained smear thoroughly with distilled water.Alternatively, place the slide on the boiling water beaker, pour malachite green onto the smear and let it remain for 5 min. Make smears from the culture on clean glass slide air dry and heat fix.Bacterial culture: Bacillus megaterium, B.Safranine or Mercurochrome (0.5% aqueous).Malachite green and methylene blue as counter stain.The information regarding spores (shape, diameter and position of endospore) is very useful for taxonomy. Spores after staining resist decolorization. Spore staining procedures make use of strong stains such as carbol fuchsin and prolonged contact with stain or the stain is poured on the smear and heated underneath. The cell wall disintegrates soon after endospore formation. Spores contain dipicolinic acid which complexes with calcium ions and thus imparts heat resistance to the spores. Endospores are metabolically inactive and resistant to heat, chemicals and harsh environmental conditions. With ordinary stain, spores remain unstained or slightly tinged with stain. The spores of bacteria do not stain as easily as vegetative cells. Out of the 10 genera that form endospores, two genera Bacillus and Clostridium are the most common.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
